Access Indicators
Sustainable Development Goal Indicator 3.b.3
2025-present
In 2025, the Health Product Access Index (Access Index) became the new SDG 3.b.3 indicator, providing a comprehensive measure of access to essential health products. Like the UHC Service Coverage Index, the Access Index is a composite index that integrates multiple tracer indicators on medicines, vaccines, diagnostics, and medical devices, which are essential for delivering effective health interventions.
This index helps assess the level of access to health products when a health intervention calls for their use. It is informed by regularly reported population-level health service coverage indicators, reflecting both the availability and equitable distribution of essential health products. The Access Index supports global efforts to monitor and improve access to health products, a critical component of achieving universal health coverage and the health-related Sustainable Development Goals.
The health product access index is based on a series of health services coverage indicators that involve the use of any or a combination of health products for their fruition.
The list of the included indicators is as follows:
- Prevalence of cervical cancer screening among women aged 30-49 years (%)
- Women accessing antenatal care (ANC) services who were tested for syphilis (%), reported
- New cases tested for RR-/MDR-TB (%)
- Tuberculosis treatment coverage
- Estimated antiretroviral therapy coverage among people living with HIV (%)
- Antenatal care attendees positive for syphilis who received treatment (%), reported
- Tuberculosis effective treatment coverage
- Women of reproductive age (aged 15-49 years) who have their need for family planning satisfied with modern methods (%)
- Measles-containing-vaccine second-dose (MCV2) immunization coverage by the nationally recommended age (%)
- Pneumococcal conjugate vaccines (PCV3) immunization coverage among 1-year-olds (%)
- HPV immunization coverage estimates among primary target cohort (9-14 years old girls) (%)
- Diphtheria tetanus toxoid and pertussis (DTP3) immunization coverage among 1-year-olds (%)
- Hepatitis B (HepB3) immunization coverage among 1-year-olds (%)
- Neonates protected at birth against neonatal tetanus (PAB) (%)
- Hib (Hib3) immunization coverage among 1-year-olds (%)
- Polio (Pol3) immunization coverage among 1-year-olds (%)
- Measles-containing-vaccine first-dose (MCV1) immunization coverage by the nationally recommended age (%)
- Population with access to an insecticide-treated bed net (ITN) for malaria protection (%)
- Prevalence of met need of assistive products (%)
These tracer indicators are meant to be indicative of access to health products and not a complete or exhaustive list of all health products required to deliver essential health services, under universal health coverage. The 19 tracer indicators were selected because they are well-established. Therefore, the index can be computed with existing data sources and does not require initiating new data collection.
Tracer indicators are measured by household surveys, facility data, sentinel surveillance systems and administrative data. In terms of values used to compute the index, values are taken from existing published sources.
The index is computed with geometric means, using the mean scores calculated for each tracer indicator group (or category of product) that is linked to the use of different health products. The index is reported on a unitless scale of 0 to 100, with 100 being the optimal value.
The mechanisms for collecting data from countries vary across the 19 tracer indicators. In many cases, a UN agency or interagency group has assembled and analysed relevant national data sources and then conducted a formal country consultation with country governments to review or produce comparable country estimates. WHO does not undertake new estimation activities to produce tracer indicator values for the health products access index; rather, the index is designed to make use of existing and well-established indicator data series to reduce reporting burden.
2018-2025
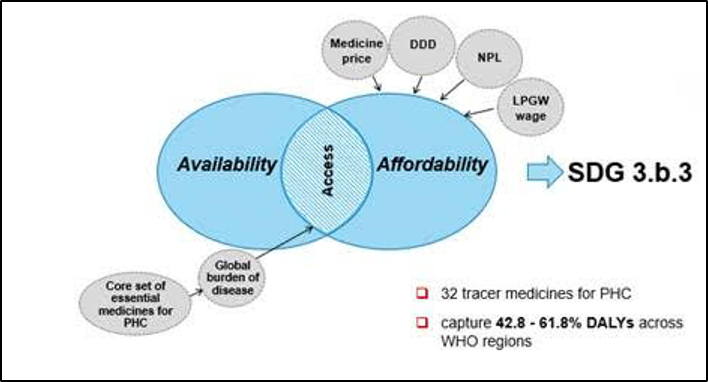
In November 2018, the United Nations Statistical Division approved the new methodology for measuring access to medicines. The Sustainable Development Goals indicator 3.b.3 is defined as the “proportion of health facilities that have a core set of relevant essential medicines available and affordable on a sustainable basis” and is intended to provide an annual estimate of a country’s access to medicines. This composite multidimensional indicator is the first to evaluate both the availability and affordability of medicines simultaneously, combining them to provide a reliable index of access. For a medicine to be considered accessible, it must be:
(i) available at a facility on the day of data collection and
(ii) affordable for an average individual who is living on the lowest government salary. If one of these conditions is not met, the medicine cannot be considered accessible
Definitions:
- Availability: A medicine is available in a facility when it is found in this facility by the interviewer on the day of data collection.
- Affordability: A medicine is affordable when no extra daily wages are needed for the lowest-paid unskilled government sector worker (LPGW wage) to purchase a daily dose treatment (DDD) of this medicine after one fulfils one’s core needs (estimated per capita per day) represented by the national poverty line (NPL).
- Access: A medicine is considered accessible when it is (i) available in a health facility and (ii) it is provided at an affordable price simultaneously.
- A core set of relevant essential medicines is a list of 32 tracer essential medicines for acute and chronic, communicable and non-communicable diseases in the primary health care setting. Each medicine in the basket is weighted according to the regional burden of disease that is treated/ cured/ controlled by this medicine.
Methodology:
To calculate an access to medicines index using the methodology below, it is essential to have nationally representative data on medicines’ availability and prices that have been collected at the facility level (e.g. data collected using the WHO MedMon mobile application, WHO/HAI Methodology, WHO Harmonised Health Facility Assessments).
Step 1: Review the core basket of medicines for a healthcare facility and select medicines which are relevant for the country and facility being analysed.
SDG 3b3 2018-2025: Core Basket
Step 2: Assign an appropriate weight to each defined medicine based on the regional burden of disease.
SDG 3b3 2018-2025: Assigning weights to medicines in core basket
Step 3: Estimate the two dimensions of access to medicines (i.e., availability and affordability) by converting both dimensions into binary variables.
SDG 3b3 2018-2025: Affordability calculation
Step 4: Combine the two dimensions of affordability and availability to produce the access to medicines index.
Step 5: Apply the appropriate weight to each medicine in the basket by multiplying the access to medicines matrix by the weight matrix.

Step 6: Aggregate data at the facility level to identify whether a facility has a core set of relevant essential medicines available and affordable.
- To achieve this, the proportion of medicines that are both available and affordable is calculated as a weighted sum of accessible medicines per facility, which is then converted into a percentage ranging from 0 to 100.
→ %
- If a facility demonstrates that 80% or more of the medicines surveyed in that facility are both available and affordable, that facility is classified as demonstrating access.
- A threshold of 80% is applied to transform the “access” variable into a binary format. This threshold is agreed upon and adopted by the WHO Global Action Plan on Non-Communicable Diseases and used as a reference in this proposed methodology.
Step 7: Aggregate data on the national level by calculating the indicator as the proportion of facilities with accessible medicines (i.e., those facilities that have attained the 80% threshold in the previous step) as a proportion of all facilities surveyed in the same country.
For further information on specific data requirements for calculating SDG 3.b.3 indicator on access to medicines, see SDG 3b3 2018-2025: Data Requirements
Data Description:
Sample Data
The following image shows an example of the estimated SDG 3.b.3 indicators on access to medicines for three randomly selected anonymised countries using the methodology described above. These estimates are based on a sample of data collected from a 2016 survey of capital cities of 19 countries.
Example of estimated SDG indicator 3.b.3 on access to medicines in three randomly selected countries
*Data was collected in the capital cities of each country, across 12-16 facilities per city.
Interpretation of the obtained results:
- In the capital city of the 1st randomly selected country (Country 1), none of the surveyed facilities provide accessible essential medicines for primary health care.
- In the capital city of the 2nd randomly selected country (Country 2), 12.5% of surveyed facilities provide accessible essential medicines for primary health care.
- In the capital city of the 3rd randomly selected country (Country 3), 69.2% of surveyed facilities provide accessible essential medicines for primary health care.
Reminder: A facility is considered to provide accessible essential medicines for primary health care if at least 80% of medicines from the defined list are available in that facility and are provided at affordable prices.
Note: An SDG 3.b.3 indicator status of “0%” indicates that none of the facilities surveyed provided at least 80% of the relevant core set of essential medicines; however, it does not indicate that none of the medicines were accessible across the surveyed health facilities in the analysed city/region/country.
Output and Outcome Indicators: Medicines and Health Technologies
OUTCOME INDICATORS
WHO’s Programme Budget outlines the priorities of the Organization, defines the targets to be delivered, and allows monitoring of their achievement on a biennial basis.
WHO has proposed two outcome indicators to measure progress on health products and technologies in its 14th General Programme of Work (GPW14).
GPW14 Outcome - Improved regulatory systems for targeted health products
OUTPUT INDICATORS
WHO has proposed the following output indicators in GPW14. These indicators capture only a proportion of WHO’s work on access to safe, effective and quality-assured health products. Other measures for outcomes, outputs and key performance indicators are reported in various departmental and project reports.
GPW14 Output Statement:
3.2.3 WHO supports countries to implement measures for better access to and use of safe, effective, and quality-assured health products
GPW14 Output Indicators:
IND1. Number of in-country registrations of prequalified products and SRA/WLA-approved products registered under the Collaborative Registration Procedure or other facilitated reliance pathway in case of emergency
IND2. Number of Member States with an established institutional development plan to improve regulatory capacity for health products based on the assessment using the WHO Global Benchmarking Tool
IND3. Number of countries with a list of essential medicines (or reimbursed medicines) developed centrally, updated within the last 5 years and grounded in the concept of the WHO Model List of Essential Medicines
For more information, please refer to:
